Anomalies of the Fetal Abdomen I: Duodenal Atresia, Limb Body Wall Complex, Cysts, Pentalogy of Cantrell
(To view a specific reference. Click on the reference number which will take you to the abstract or article.)
Duodenal Atresia: Information
Page Links: Fetal Bowel Obstruction, Meconium Peritonitis, Epidemiology, Relationship to Other Anomalies, Ultrasound Considerations, MRI, Non-Ultrasound Methods: Small Bowel Abnormalities, Associated Malformations, Neonatal Care
Fetal Bowel Obstruction
The presence of dilated bowel loops suggests fetal bowel obstruction.
Meconium Peritonitis
Following fetal bowel rupture, the findings suggestive of meconium peritonitis are: ascites, intra-abdominal calcifications, meconium cysts, dilated loops of bowel, and polyhydramnios.
Presently, there are no interventions for fetal gastrointestinal anomalies, but early detection allows planned delivery, appropriate resuscitation, and neonatal surgical intervention. [1]
Epidemiology
In a series of 99 patients with small intestinal atresia (SIA), 25.8% were associated with chromosomal aneuploidy. [2] In an epidemiological study of small intestinal atresia in Europe, among 1044 singleton cases, 215 or 20.6% were associated with a chromosomal abnormality and there were no significant prevalence variations over time. [3] The prevalence per 10,000 births was 1.6 for small intestinal atresia, 0.9 for duodenal atresia and 0.7 for jejunoileal atresia (JIA). [3]
Relationship to Other Anomalies
Duodenal atresia was noted in 4 of 94 consecutive fetuses with Down syndrome. [4] Duodenal atresia, fetal hydrops and omphalocele are the most specific single ultrasound anomalies associated with an abnormal karyotype. [5] Duodenal atresia is the malformation most commonly associated with an abnormal karyotype detected in late pregnancy. [6]
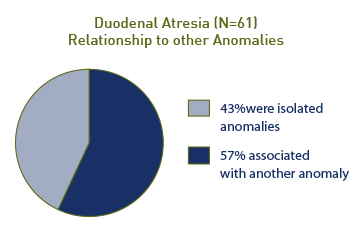
In a series of 61 patients with duodenal atresia, 35 (57%) were associated with another congenital abnormality and 26 (43%) were isolated anomalies.
Ultrasound Considerations
Among those who were diagnosed prenatally, 71% (21 out of 29) had associated malformations. [7] Duodenal atresia was suspected on the 20 week ultrasound in 29 of 33 cases, and there were 4 false positives (1 normal and 3 with high jejunal atresia). [7]
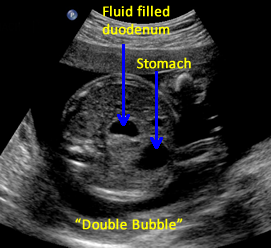
The diagnosis of duodenal atresia has been made as early as 14 weeks gestation. [8] About 87% of fetuses with congenital duodenal obstruction will demonstrate the “double bubble” sign, and duodenal “windsocks” were observed in 3 fetuses classified as normal that went on to have duodenal obstruction. [9] In 5 of 24 cases, the double bubble sign was noted prior to 20 weeks, and associated anomalies included biliary atresia and malrotation. (Associated malformations were noted in 4 of 18 (22%) patients with a normal karyotype). [9]
The double bubble sign may be confused with other intra-abdominal abnormalities such as duodenal duplications, which are characterized by a lack of polyhydramnios, failure to consistently demonstrate the abnormality, and the presence of normal distal bowel. [10] In addition, normal fetuses may exhibit a transient double bubble sign. [11]
MRI
MRI is useful in a number of fetal gastrointestinal malformations, particularly the more complex ones [12], and inversion mode used in 3-D ultrasound is useful in defining irregular or cystic structures containing fluid. [13]
Non-Ultrasound Methods: Small Bowel Abnormalities
Other non-ultrasound methods have been used to assess small bowel abnormalities. Amniotic fluid gamma-glutamyl-transpeptidase (GGTP) and intestinal alkaline phosphatase (iALP) were evaluated from the amniotic fluid in 55 patients with fetal gastric or small bowel dilatation. Abnormal amniotic fluid enzyme activities were confirmatory of obstructive pathology. [14]
Associated Malformations
A number of other associated malformations have been reported with duodenal atresia, which include: congenital diaphragmatic hernia [15], VACTERL syndrome [16] as well as cardiac defects, annular pancreas, and intestinal malrotation. [17]
Neonatal Care
The operative mortality is < 1% for duodenal atresia. The survival rate improves if the atresia is distal. [18]
Multiple atresias increase mortality (57%), while “apple peel” atresia has a mortality of 71%. The following mortalities were reported: atresia with meconium ileus (65%), meconium peritonitis (50%), and gastroschisis (66%). [18]
Most deaths in patients with congenital duodenal obstruction are associated with congenital anomalies. [19]
Duodenal Atresia: Images
Above. Duodenal atresia. 21 5/7 weeks gestation. Large fetal stomach “bubble.”
Above. Duodenal atresia. 21 5/7 weeks gestation. “Double bubble” sign.
Above. Duodenal atresia. Fetal connection between the stomach and the duodenum via the pylorus.
Duodenal Atresia: Video
Above. Duodenal Atresia. 21 5/7 weeks gestation. Large fetal stomach.
Above. Duodenal Atresia. 21 5/7 weeks gestation. “Double bubble” sign.
Above. Duodenal Atresia. 21 5/7 weeks gestation. “Double bubble” sign.
Above. Duodenal Atresia. 30 5/7 weeks gestation. Trisomy 21 fetus with duodenal
atresia and AV canal defect.
Limb Body Wall Complex: Information
Page Links: Definition, Incidence, Classification, Etiology, Risk Factors, Outcome, Management, Differential Diagnosis, References
Definition
Limb body wall complex (LBWC), which is also known as body stalk anomaly, is a rare, congenital defect.
Diagnosis
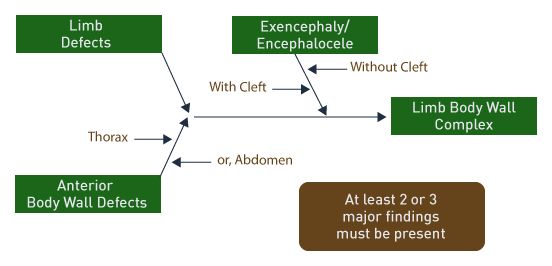
At least two of three characteristics are necessary for diagnosis of limb body wall complex [20]:
1. Limb defects.
2. Anterior body wall defects.
3. Exencephaly or encephalocoele with/without facial clefts.
The limb defects vary from absence of a limb to positional changes and club foot. The defects in the abdomen and chest are large and are sometimes combined. The anterior abdominal wall defects differ from those seen with typical gastroschisis or omphalocele. Multiple other anomalies may be present, which include: short umbilical cord, single umbilical artery, severe kyphoscoliosis, external genitalia, and anomalies of the heart, colon, and kidneys.
Incidence
The incidence of limb body wall complex is reported as .33 per 1000 live births1 and 1 in 7623 cases screened between 10 and 14 weeks gestation.2 In the latter group, a major abdominal wall defect, severe kyphoscoliosis, and a short umbilical cord were noted. A history of use was reported for cigarette (50%), alcohol (50%) and marijuana (30%) in women with LBWC. In 40%, there was a history of a previous infant with a congenital anomaly and one patient in the series delivered two consecutive male infants with LBWC.
Classification

Above. Classification of limb body wall defects is based upon potential etiology.3 Abnormal development of each of the 4 embryonic folds may be factors responsible for these malformations.
Another potential classification system is based upon the presence or absence of amniotic bands. [21] Body stalk anomalies with and without amniotic bands may be different entities. [5]
Etiology

A number of etiologies have been proposed. Among them is an abnormality of early embryonic folding. [22] This etiology also serves to classify the malformation. The embryo folds into 4 planes and in so doing allows the normal development of the peritoneal cavity and separation from the umbilical cord.
The limb body wall malformations result from a malfunction of the ectodermal placodes involving the early embryonic folding process. [23]
Others have observed and propose a vascular etiology. [24]
A combination of these mechanisms also include: early amnion rupture, vascular disruption, and embryonic dysgenesis. [25]
Risk Factors
Limb body wall anomalies are unlikely to be associated with karyotype abnormalities. [26]
The fetal karyotype was normal in 12 fetuses studied at 10 to 14 weeks gestation. [27]

Above. The factors cited above increase the risk for limb body wall complex. [28],[29]
Outcome
Limb body wall complex is most often considered a lethal anomaly. Detection by the end of the first trimester is possible. [30]
Management
If limb body wall complex is suspected, targeted ultrasound is indicated with referral to a tertiary care center. While the recurrence risk is low, genetic counseling is indicated.
Limb Body Wall Videos
Above. Limb body wall complex. 22.0 weeks. The heart is
displaced inferiorly into a large anterior abdominal wall defect.
Above. Limb body wall complex. 22.0 weeks. Again, there is
a large anterior wall defect with viscera attached to the placenta.
Above. Limb body wall complex. 20.1 weeks. The spine
is angulated and distorted. It is difficult to obtain
longitudinal views of the spine.
Above. Limb body wall complex. 20.1 weeks. The
umbilical cord is short and contains a single artery.
Above. Limb body wall complex. 22.0 weeks. This is another
demonstration of the herniated viscera’s attachment to the placenta.
Intra-abdominal Cysts and Masses: Information
Intra-abdominal cysts are a varied and sometimes difficult to define on an antenatal ultrasound. Their origins are diverse due to the multiplicity of developmental histories including that of the fetal bowel, genitourinary system, and organ development such as the liver, pancreas, and adrenal gland. In addition, cysts or masses are less likely to be seen at the 20 week scan. [31]
A number of intra-abdominal cysts resolve on postnatal exam and a nonspecific diagnosis is made in a high percentage of cases. In a study of 55 patients with a prenatal diagnosis of intra-abdominal cysts, 24% (13) resolved and 29% (16) required surgery. [32]
A nonspecific diagnosis was made in 47% (26) and a specific diagnosis was made in 53% (29).
Among those with a specific diagnosis, 43% (12) were confirmed postnatally and 57% (17) were not confirmed postnatally. [2]
Others support the supposition that a definitive diagnosis frequently is not made until the neonatal period and most intra-abdominal cysts do not alter obstetric management. [33]
Abdominal cysts are frequently misdiagnosed and are among the most common false positive results occurring with fetal structural anomalies. [34] For example, ovarian cysts account for a false positive rate as high as 17%, which is higher than the overall false positive rate for fetal anomalies. [35]
References
Limb Body Wall
Pentalogy of Cantrell: Information
Page Links: Definition/Prevalence, Classification, Etiology, Genetics, Associated Malformations, Outcome, Differential Diagnosis, References
Definition/Prevalence
Pentalogy of Cantrell is a rare congenital malformation, which was first described in 1958. Classically, it consists of 5 findings. [36] The prevalence is reported as 1 in 65,000 births. [37]
Classification
Pentalogy of Cantrell can be classified into complete and incomplete forms. [38] A fewer number of defects in the malformation have been reported. Prognosis depends upon the number and severity of the defects. [3]
Etiology
Above. The proposed etiology by Cantrell is developmental failure of the lateral mesoderm with impaired migration of the mesodermal fold. [39] The transverse septum fails to develop and myocardial development is also impaired. [40]
Genetics
Above. Genetic Considerations for Pentalogy of Cantrell
Trisomy 18 has been reported with the critical features of pentalogy of Cantrell, but also with additional manifestations. [41] Other cases of trisomy 18 associated with pentalogy of Cantrell also exhibit hypogenesis of the corpus callosum, Dandy-Walker variant, ventricular septal defect, dextrocardia, and patent ductus arteriosus. [42]
Goltz-Gorlin syndrome affects body parts of meso-ectodermal origin. [43] It has been described as having features of pentalogy of Catrell in a number of case reports. A point mutation in PORCN (c.727C>T) in a case of molecularly confirmed Goltz-Gorlin syndrome and pentalogy of Cantrell has been reported, which suggests that, in some patients, pentalogy of Cantrell can be caused by a PORCN mutation. [8] Others report an association between the coding gene sequence of PORCN and a number of mutations in the Goltz-Gorlin syndrome. [44] A search for the PORCN mutation seems reasonable in cases of pentalogy of Catrell, especially when there are limb abnormalities. [45]
Adding to the potential genetic basis of pentalogy of Cantrell, 3 consecutively-born brothers were born with the syndrome. [46]
Associated Malformations
Above. A wide variety of associated defects have been reported with Pentalogy of Cantrell. [47],[48],[49],[50] These include a bilateral cleft lip and palate. [12]
Cardiac defects include patent ductus arteriosus, atrial and ventricular septal defects. [12] tricuspid atresia type IIc [13], and tetralogy of Fallot. [15]
Rarer expressions of this disorder include asymmetric kidneys. [13]
Previously unreported associations include bilateral intra-abdominal testis and lymphocytic meningitis in one case, and adreno-hepatic fusion, anterior myeloschisis, Meckel diverticulum, multiple accessory spleens, and renal agenesis in another case. [14]
Gallbladder agenesis and polysplenia have also been reported. [15]
Outcome
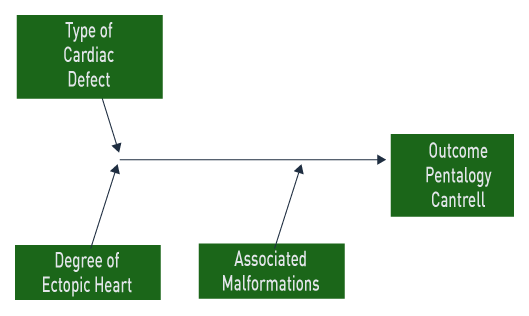
Above. In pentalogy of Cantrell, the outcome depends upon the type of cardiac malformation, the presence of an ectopic heart, and associated malformations. In one series, the overall survival was 4 in 21 patients and no infants survived in the presence of an ectopic heart. [51]
In another earlier series of 10 cases of pentalogy of Cantrell, there were no survivors. [52]
Above. Successful 2 stage repair of pentalogy of Cantrell with tetralogy of Fallot has been reported. [53] In 13 patients with conotruncal defects and either thoracoabdominal ectopia cordis (n=9) or thoracic defects (n=4), survival after repair was reported in 4 of 8 with thoracoabdominal ectopia cordis and 1 of 2 with thoracic defects. [54]
Others report successful operative outcomes for pentalogy of Cantrell in patients with a ventricular septal defect (VSD) and a left ventricular diverticulum. [55]
Postoperative care in patients undergoing surgical repair of cardiac defects is complicated in some instances by the need for prolonged ventilator support, [56] while circulatory and ventilator management of such patients is complex. [57]
Successful one stage repair of pentalogy of Cantrell has been reported using a technique of plicating the eventrated diaphragm and returning the heart to the thoracic cavity by fixing the pericardium to the ventral side of the diaphragm. [58]
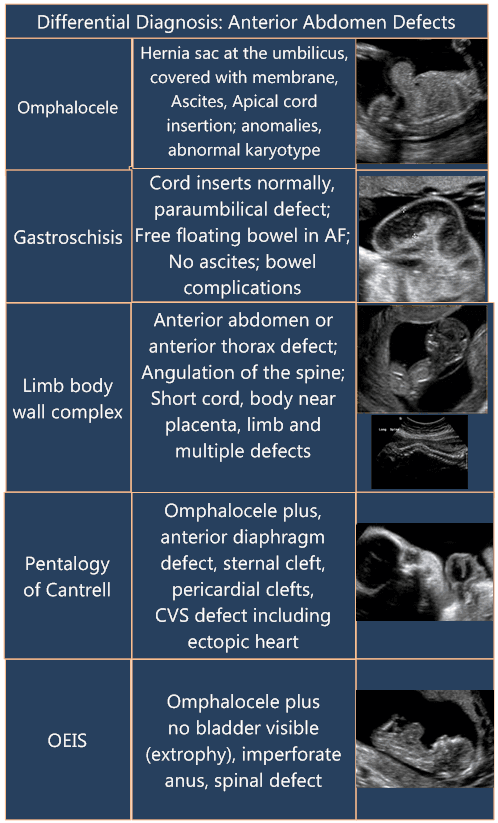
Differential Diagnosis
Pentalogy of Cantrell: Images
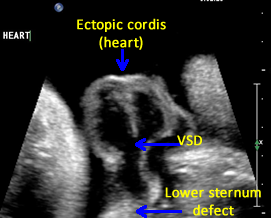

Above. Pentalogy of Cantrell. Case 1. 34 6/7 weeks gestation. Mid-line lower sternum defect with ectopic cordis.
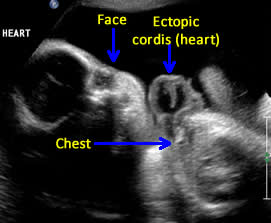

Above. Pentalogy of Cantrell. Case 1. 34 6/7 weeks gestation. Pentalogy of Cantrell showing relationship of ectopic heart to fetal face.
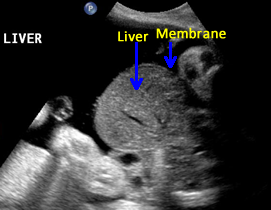
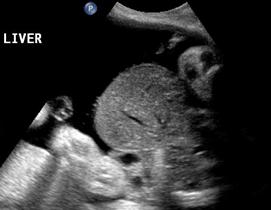
Above. Pentalogy of Cantrell. Case 1. 34 6/7 weeks gestation. Pentalogy of Cantrell showing associated omphalocele containing fetal liver. Note the thin membrane covering of the omphalocele defect.
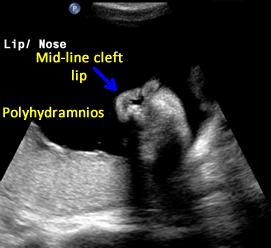

Above. Pentalogy of Cantrell. Case 1. 34 6/7 weeks gestation. Pentalogy of Cantrell showing cleft lip, another mid-line defect.
Above. Pentalogy of Cantrell. Case 1. 34 6/7 weeks gestation. Pentalogy of Cantrell showing sagittal view of fetal profile with the possibility of frontal bossing. Note the large mid-line cleft.
Above. Pentalogy of Cantrell. Case 1. 34 6/7 weeks gestation. Pentalogy of Cantrell showing additional malformations. Note brachycephaly and the obliteration of the posterior fossa.
Pentalogy of Cantrell: Video
Above. Pentalogy of Cantrell. 34 6/7 weeks gestation.
Ectopic heart secondary to lower sternum defect.
Above. Pentalogy of Cantrell. 34 6/7 weeks gestation.
Ectopic heart with VSD (ventricular septal defect).
Above. Pentalogy of Cantrell. 34 6/7 weeks gestation.
Note omphalocele with hepatic vessels. The cord insertion
is noted at the end of the video.
Above. Pentalogy of Cantrell. 34 6/7 weeks gestation.
The bowel and liver are outside of the abdomen.
-
Abstract: PMID: 16626900 -
Abstract: PMID: 17994616 -
Abstract: PMID: 22267397 -
Abstract: PMID: 2143275 -
Abstract: PMID: 1481814 -
Abstract: PMID: 8650127 -
Abstract: PMID: 19551391 -
Abstract: PMID: 7619097 -
Abstract: PMID: 10955559 -
Abstract: PMID: 9430213 -
Abstract: PMID: 8809900 -
Abstract: PMID: 15136891 -
Abstract: PMID: 15661951 -
Abstract: PMID: 7899271 -
Abstract: PMID: 8807765 -
Abstract: PMID: 2681660 -
Abstract: PMID: 8337875 -
Abstract: PMID: 11028122 -
Abstract: PMID: 1555060 -
Abstract: PMID: 11527200 -
Abstract: PMID: 10797417 -
Abstract: PMID: 17886024 -
Abstract: PMID: 11527200 -
Abstract: PMID: 18699998 -
Abstract: PMID: 11186176 -
Abstract: PMID: 10797417 -
Abstract: PMID: 9476328 -
Abstract: PMID: 12420845 -
Abstract: PMID: 12749386 -
Abstract: PMID: 11186176 -
Abstract: PMID: 18567068 -
Abstract: PMID: 18443802 -
Abstract: PMID: 12780436 -
Abstract: PMID: 17154189 -
Abstract: PMID: 16567202 -
Abstract: PMID: 13592660 -
Abstract: PMID: 21448392 -
Abstract: PMID: 21448392 -
Abstract: PMID: 13592660 -
Abstract: PMID: 15728634 -
Abstract: PMID: 3232702 -
Abstract: PMID: 18947004 -
Abstract: PMID: 21484999 -
Abstract: PMID: 21472892 -
Abstract: PMID: 19586929 -
Abstract: PMID: 1554024 -
Abstract: PMID: 21448392 -
Abstract: PMID: 18782955 -
Abstract: PMID: 15127772 -
Abstract: PMID: 14694384 -
Abstract: PMID: 21652132 -
Abstract: PMID: 3066921 -
Abstract: PMID: 8215683 -
Abstract: PMID: 8901716 -
Abstract: PMID: 17885784 -
Abstract: PMID: 19322603 -
Abstract: PMID: 19921368 -
Abstract: PMID: 8840759