Anomalies of The Fetal Abdomen III: Umbilical Vein Varix, Calcifications, Meconium Peritonitis, Plus Sacrococcgeal Teratoma (2.0 CME Credits)
Intra-abdominal Umbilical Vein Varix: Information and Imaging Considerations
(To view a specific reference. Click on the reference number which will take you to the abstract or article.)
Page Links: Definition, Diagnosis, Outcome, Other Presentations, Management
Definition
Intra-abdominal umbilical vein varix is an aneurysmal dilatation of the umbilical vein. It is associated with an increased fetal death rate and chromosomal abnormalities. Umbilical vein varix is defined as a width of the intra-abdominal portion of the umbilical vein that is > 2 standard deviations in size for its gestational age. The umbilical vein varix is both focal and aneurysmal. [1] Approximately 100 cases have been reported in the literature. [2]
Diagnosis
Elevation of maternal serum alpha-fetoprotein (AFP) and mesenchymal dysplasia has been reported in association with umbilical vein varix. [3]
The umbilical vein varix is typically an ovoid, elongated, fluid-filled mass that is obliquely oriented between the abdominal wall and the edge of the liver. [4] Color Doppler confirms venous flow and is diagnostic for the lesion. Three-dimensional power Doppler (3DPD) ultrasound may be of additional value. [5] Umbilical artery waveform notching may be a predictor for adverse events in instances of umbilical cord abnormalities. [6]
Outcome
Some studies in small series suggest reassuring outcomes. [7] However, other larger reviews confirm the possibility for adverse events and a poor outcome.
In a review of 91 cases of intra-abdominal vein varix, additional anomalies were detected in 31.9%. In intra-abdominal vein varix, the cardiovascular system is affected most often followed by fetal hydrops and fetal anemia. [8] Karyotype abnormalities occurred in 9.9% of cases. 13% of affected fetuses suffered perinatal deaths and only 59.3% of cases had a normal outcome. Even in cases of isolated umbilical vein varix, intrauterine deaths occurred in 8.1%. Early diagnosis (< 26 weeks) was associated with higher morbidity. [8]
In another study of 25 patients, only 48% had normal outcomes, 13% delivered prematurely, and 35% had structural malformations. [9] In a third study of 26 fetuses, there were other malformations in 19.2%. One had trisomy 21 and one had fetal growth restriction. [10]
Other Presentations
AV malformations involving the intra-hepatic portion of the umbilical vein have been reported with trisomy 21. [11] Umbilical vein varix has also been reported in association with Klippel-Trenaunay-Weber (KTW) syndrome, which is a rare vascular malformation that includes soft-tissue hypertrophy, varicose veins, cutaneous hemangiomas, and arteriovenous fistulas). [12]
Fetal anemia may be another associated finding. Schistocytic (fragmented red blood cells) anemia can be assessed by the peak systolic velocity of the middle cerebral artery. [13] Postnatally, the varix may be confused with an incarcerated umbilical hernia. [14]
Management
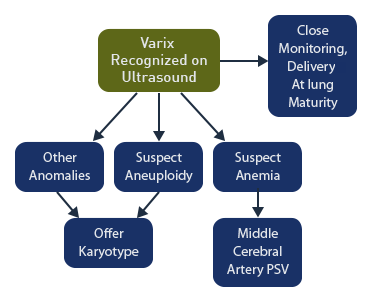
Prenatal recognition of the varix allows for assessment of other malformations including karyotype abnormalities. If other malformations or makers of aneuploidy are present, the karyotype testing should be offered. If fetal anemia is suspected, the peak systolic velocity of the middle cerebral artery should be assessed. Some authors recommend close fetal monitoring and delivery at lung maturity. [15] However, intrauterine demise can occur despite close fetal surveillance. [16]
Intra-abdominal Umbilical Vein Varix: Images
Above. Umbilical vein varix. Case 1. Gestational age 33 3/7 weeks. Transverse scan. Anechoic area within the fetal liver near the umbilical cord insertion site. The diameter is 14 mm.


Above. Umbilical vein varix. Case 1. Gestational age 33 3/7 weeks. Oblique scan. Anechoic area within the fetal liver near the umbilical cord insertion site.


Above. Umbilical vein varix. Case 1. Gestational age 33 3/7 weeks. Color Doppler demonstrates flow and confirms the diagnosis.


Above. Umbilical vein varix. Case 1. Gestational age 33 3/7 weeks. Doppler velocimetry demonstrates mildly pulsatile flow near the varix.
Intra-abdominal Calcifications: Information and Imaging Considerations
The differential diagnosis for calcifications within the fetal abdomen includes [17]: meconium peritonitis, infections (cytomegalovirus, toxoplasmosis, and varicella), and neoplasms (teratoma, neuroblastoma, hemangioma, and hepatoblastoma). The infectious origins favor calcifications in the liver and spleen. Many rare conditions are also associated with fetal intra-abdominal calcifications.
In meconium peritonitis, isolated intra-abdominal calcifications have excellent outcomes [18] However, if the calcifications are associated with ascites, pseudocyst, or bowel dilatation, the prognosis is worse than that of a group with intra-abdominal calcifications only. [2]
Meconium peritonitis is the most common cause of fetal abdominal calcifications. In one study identifying fetuses with abdominal calcifications, meconium peritonitis was defined as “simple” if there were no bowel abnormalities and “complex” if bowel abnormalities were present. [19] Dilated bowel loops, meconium cysts, ascites, and polyhydramnios were features of those with complex meconium peritonitis. [3] In addition, fetal hydrops is associated with meconium peritonitis in some ultrasound studies. [20]
Among 194 cases of newborns with meconium peritonitis, “the radiographic features could be categorized as pneumoperitoneum with intestinal obstruction (adhesions) and calcification (35 cases), intestinal obstruction with calcification (143 cases), intestinal obstruction with no radiographically visible calcification (5 cases), and calcification alone (11 cases).” [21]
In a small series of patients with meconium peritonitis, 5 of 12 (42%) had cystic fibrosis and the calcifications were intraperitoneal. [22] However, the absence of calcifications in cases with other findings suggestive of meconium peritonitis, such as ascites and polyhydramnios, favors a diagnosis of cystic fibrosis. [6]
In cystic fibrosis, calcifications are more likely to occur within the bowel wall (intramural) than within the bowel lumen (intraluminal) or the serosa, and the pattern of calcifications varies from small specks to denser and more curvilinear calcifications. [23]
Other causes of intra-abdominal calcification include anorectal anomalies, rectourethral fistula, and intraluminal calcified meconium. [24],[25] Intraluminal calcifications may be due to a communication between the urinary and gastrointestinal tracts. [26]
Enterolithiasis may be a component of the VACTERL (vertebral defects, imperforate anus, cardiac anomalies, tracheoesophageal fistula, renal and limb) anomalies. [27] The imperforate anus in that syndrome is associated with recto-urinary fistula and the stasis of urine leads to intraluminal calcifications. [28]
In neonates, calcifications have been observed in rectal atresia without fistula, jejunal atresia, and stenosis of the distal small bowel associated with long segment Hirschsprung’s disease. [29]
Intraluminal calcifications are seen in total colonic aganglionosis, and are manifested in small punctate calcific densities in the lower abdomen in association with bowel obstruction. They are often indicative of microcolon. [30] This type of calcification is also seen in small intestinal atresia and stenosis. [14]
Other rare causes of calcification are: fetus-in-fetu [31], hydrometrocolpos secondary to imperforate hymen [32], and neonatal small left colon syndrome (NSLCS) with meconium peritonitis. [33]
Calcified duodenal duplication cysts are seen in the upper abdomen and are spherical in shape. [34]
Ileal atresia may be associated with ileo-ileal intussusception at the level of the ileocecal valve. The intussusception is manifested in intestinal loops with high echogenicity and a “target-like” appearance. [35]
Neonatal perspective
Abdominal calcifications may be “incidental” findings on a neonatal X-ray, that correlates with an in-utero perforation. This can be useful information if the neonate has feeding intolerance. With cystic fibrosis now a part of newborn screening, neonatologists may be less aggressive in defining cystic fibrosis immediately after birth. However, a sweat chloride test is commonly recommended when the child is older regardless of the screen result, unless another reason is found to explain the meconium peritonitis.
Intra-abdominal Calcifications: Images

Above. Diffuse intra-abdominal calcifications. Case 1. 20 weeks gestation. Possibilities include: fetal infection such as toxoplasmosis, herpes simplex, varicella, CMV, and parvovirus. Meconium peritonitis is always a possibility, but is usually associated with bowel dilation and ascites.
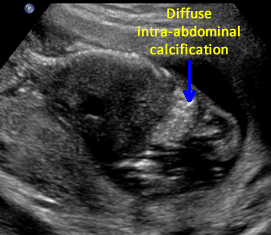

Above. Diffuse intra-abdominal calcifications. Case 1. 20 weeks gestation. Diffuse intra-abdominal calcifications. Echogenicity is equal to bone in this case.

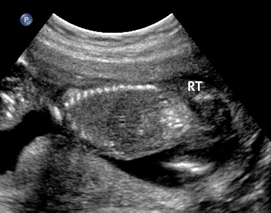
Above. Diffuse intra-abdominal calcifications. Case 1. 20 weeks gestation. In this case, the calcifications do not appear to involve the fetal liver.
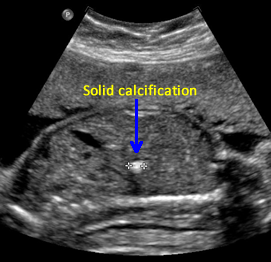

Above. Calcification. Case 2. 23 weeks gestation. Single echogenic focus in the liver with echogenicity equal to bone. No adverse clinical outcome was noted.
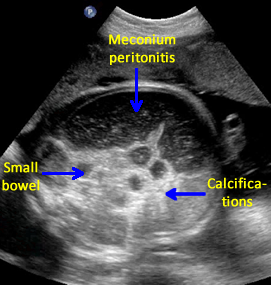

Above. Calcifications due to meconium peritonitis. Case 2. 36 6/7 weeks gestation. Note dilated small bowel and fetal ascites. Meconium peritonitis is one of the most frequent causes of calcifications within the fetal abdomen.

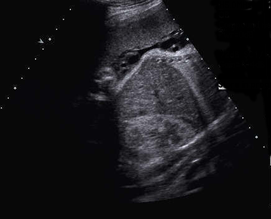
Above. Heterogeneous hepatic tumor mass with areas of calcification. Case 3.
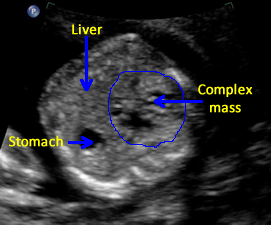

Above. Heterogeneous hepatic tumor mass with areas of calcification. Case 3.
Intra-abdominal Calcifications: Video
Above. Diffuse intra-abdominal calcification. Etiologies have been discussed previously. The calcification is less focal and intense. It may represent swallowed fetal blood.
Above. Califications are more focal. Possibilities include fetal infection. There is no bowel dilatation to suggest meconium peritonitis.
Meconium Peritonitis: Information
Meconium peritonitis (MP) was first described by Carl von Rokitansky in 1838. [36] It is the result of a rupture of the fetal bowel prior to birth, which causes meconium to spill into the peritoneal cavity. Pseudocysts are loculations of fluid, which occur as a result of the inflammatory response to meconium.
Risk factors for fetal bowel obstruction and meconium peritonitis include cystic fibrosis and every type of congenital bowel malformation such as intestinal atresia, fecal (meconium) obstruction, and other intra-abdominal malformations. The presence of twins, particularly from in vitro fertilization, and maternal hepatitis may be predisposing factors. (See below)
In a large series of fetal gastrointestinal malformations, approximately 11% were related to meconium peritonitis. [37]
A classification scheme for meconium peritonitis in utero is as follows [38]: Type I (massive meconium ascites), Type II (giant pseudocyst), and Type III (calcification and/or small pseudocyst). All three types have been reported with abdominal calcifications and/or polyhydramnios. [3]
In another series of 19 fetuses, “mean gestational age at diagnosis was 29.1 +/- 5.3 weeks and mean follow-up before delivery was 7.5 +/- 6.2 weeks.” [39] Type I (meconium ascites) was found in 26.3% of cases, Type II (large pseudocyst) in 15.8% of cases, and Type III (calcifications, smaller ascites, and/or shrinking pseudocyst) in 57.9%. [4]
In another report, the finding on prenatal ultrasound of ascites, bowel calcifications, and dilated or hyperechogenic bowel loops allowed accurate diagnosis in 3 of 7 cases (43%). [40]
In some studies, meconium peritonitis diagnosed prior to birth is associated with a high frequency of fetal ascites, calcifications, dilated bowel loops, and pseudocysts. The following ultrasound findings were demonstrated [41]:
Fetal ascites (100%).
Intra-abdominal calcifications (93%).
Dilated bowel loops (57%).
Pseudocysts (29%).
Polyhydramnios (50%).
Among 17 patients, prenatal ultrasound predicted the need for neonatal surgery when there is persistent pseudocyst, ascites, and dilated bowel loop. [42]
Intussusception is a rare event, but intestinal loops with high echogenicity and a target-like appearance is suggestive of intussusception. [43]
Maternal fulminant hepatitis B [44] and maternal hepatitis A [45] have been reported in separate cases of meconium peritonitis.
A number of reports of meconium peritonitis in twin gestations suggest but do not confirm a predisposing association. Meconium peritonitis has been noted in both twins of a twin pregnancy, each fetus with varying severity of the disease. [46] Meconium peritonitis also has been reported in other twin gestations [47] and after IVF which resulted in a twin gestation. [48]
MRI is useful in the evaluation of large meconium pseudocysts identified during the third trimester. [49] MRI is of particular value in assessing displaced organs as a result of an expanding pseudocyst [50] and it can be used to easily differentiate pseudocysts from enteric cysts. In addition, normal bowel is readily identified and assessed with MRI. [51]
In cases of massive ascites, repetitive decompression parancentesis may prolong gestation and improve outcome. [52],[53]
Following birth, surgical therapy may involve peritoneal drainage followed by laparotomy. [54]
Meconium Peritonitis: Images

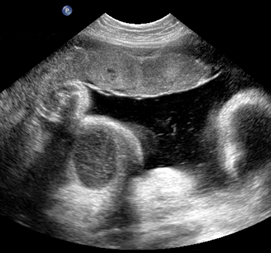
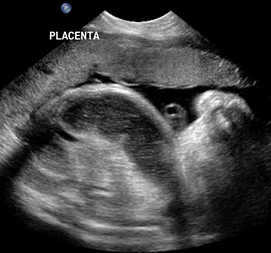
Above. Meconium peritonitis. Case 1. 36 6/7 weeks gestation. Transverse small bowel. Note small bowel dilatation and polyhydramnios.
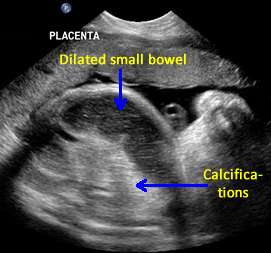
Above. Meconium peritonitis. Case 1. 36 6/7 weeks gestation. Longitudinal small bowel. Note small bowel dilatation and mesenteric calcifications.


Above. Meconium peritonitis. Case 1. 36 6/7 weeks gestation. Again, note small bowel dilatation and mesenteric calcifications.

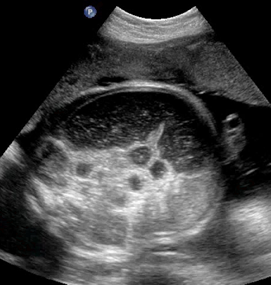
Above. Meconium peritonitis. Case 1. 36 6/7 weeks gestation. Again, note small bowel dilatation and mesenteric calcifications.
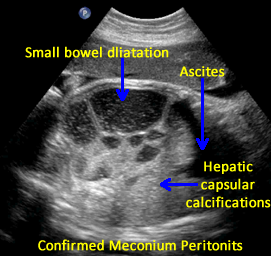

Above. Meconium peritonitis. Case 1. 36 6/7 weeks gestation. Note hepatic capsular calcifications and the presence of fetal ascites.
Fetal Colon
Normal Measurements
The fetal colon can be visualized as early as 22 weeks as a tubular structure around the perimeter of the fetal abdomen. [55] The colon demonstrates no peristalsis and the diameter is related linearly to gestational age, reaching an 18 mm maximum diameter at term. [1] The diameter of the descending colon as a function of gestational age has a sigmoid curve. The mean values and the 95% limits have been described. [56] Overall, the fetal small bowel rarely exceeds 6 mm and the colon lumen rarely exceeds 23 mm, and haustra folds are frequently demonstrated. [57]
Screening Ultrasound
Screening of the fetal abdomen usually is not done in a systematic manner. Ascites, dilatation of bowel, or fluid filled cysts are detected in the fetus, but esophageal atresia and anorectal malformations are frequently missed. [58] Coronal views of the esophagus, small bowel, and colon may be beneficial as well as median cuts of the rectum.
Recently, two scanning techniques to assess the fetal anus have been proposed [59]:
1. A tangential scan posterior to the external genitalia of the fetal perineum. The anus is seen as a hypoechoic ring which represents the wall of the anal canal with a central echogenic dot representing the lumen.
2. A coronal scan of the fetal pelvis is useful to assess the anal canal in continuity with the rectum.
Anal Atresia
In cases of imperforate anus or anorectal atresia, dilatation of the fetal colon can be seen as early at 12 weeks. [60],[61] In some cases of anal atresia, the colon may be transiently dilated as early as 16 weeks and then revert to a normal colonic appearance by 20 weeks. [62]
In many cases of anorectal atresia, associated malformations are present. In one study, eleven of 12 fetuses (92%) with anorectal atresia had malformations related to the VACTERL syndrome (vertebral defects, anal atresia, tracheoesophageal fistula with esophageal atresia, radial and renal dysplasia, and limb malformations). In most cases, ultrasound is able to define one or more of these malformations. [63]
Imperforate anus may be seen with intraluminal calcifications in dilated loops of intestines in the presence of a urinary tract abnormality [64], and other rare combinations of dilated large bowel loops, echogenic structures within the lumen, and oligohydramnios suggest anal-urethral atresia with vesico-rectal fistula. [65]
Colonic Atresia
The transverse colon may be dilated in cases of colonic atresia [66], and colonic atresia with obstruction and rupture may present as isolated fetal ascites. [67]
Syndromes
Notochord syndrome is a combination of a spinal cystic lesion with a protruding segment of bowel, and variants may include imperforate anus with recto-urethral fistula. [68]
Dilatation of the sigmoid colon and the finding of a beak-like nose is suggestive of the rare autosomal recessive disorder, Johanson-Blizzard syndrome. [69]
Hirschsprung’s disease
Prenatal diagnosis of Hirschsprung’s disease is suggested by dilated bowel loops, increased abdominal circumference, and mild polyhydramnios. [70]
Megacystis-microcolon-intestinal hypoperistalsis syndrome (MMIHS)
Megacystis-microcolon-intestinal hypoperistalsis syndrome (MMIHS) is a rare, usually lethal, autosomal recessive disorder characterized by dilatation of the urinary tract, and fetal colon with normal amniotic fluid volume. [71] The syndrome has also been reported in association with trisomy 18. [72] Microscopic findings indicate a visceral myopathy in the bowel and bladder of infants affected with MMIHS, [73] and therefore, the obstruction of the bladder and intestines is functional. [74] Most cases occur in females. [75]
A number of cases of MMIHS have been detected in utero. Ultrasound findings include persistent gastric dilatation. Pathologic findings include: megacystis, bilateral hydronephrosis and megaureters, short bowel, microileum, microcolon, and malrotation of the intestines. [76]
MRI and enzyme analysis help identify obstruction and define MMIHS. [77] In the ultrasound assessment, the dilatation of the fetal bladder obscures the dilatation of the colon, but the diagnosis is suggested by a normal sodium and chloride from the bladder aspirate, normal amniotic fluid volume, and findings of colon dilatation. [78] Clinical presentation of MMIHS may include a rapidly progressive bilateral hydronephrosis and bladder dilation but normal amniotic fluid volume, [79] and in some cases, polyhydramnios does not appear until the third trimester. [80]
A positive family history may be useful in cases of a cystic structure in the pelvis, enlarged stomach, dilated bowel, and prominent renal pelvis [81] MMIHS may be present in combinations with other renal malformations such as bilateral renal duplicated collecting systems. [82]
Chloride Diarrhea
Congenital chloride diarrhea is a rare hereditary syndrome [83] characterized on ultrasound by dilated loops of bowel, fluid within the bowel, and polyhydramnios. MRI may be particularly useful if the condition is suspected since distinction is possible between fluid within the bowel and meconium. [84] Prenatal diagnosis is warranted because of the need for identification and neonatal treatment.
Similarly to chloride diarrhea, microvillus inclusion disease also may present with dilated bowel loops and polyhydramnios. In this condition, molecular analysis is possible and may guide genetic counseling. [85]
Duplications
Duplication of the transverse colon is unusual for bowel duplication. [86] Only 13% of duplications occur in the colon, while the most common site is the ileum. [87] A significantly dilated large bowel with hyper-peristalsis suggests complete duplication of the colon. [88] Cystic duplication of the colon may be confused with an ovarian cyst, which is a more common entity. [89] Unusual presentations include cystic duplication of the descending colon with sigmoid colon enteric communication [90], and combined duplication of the bladder and bowel from ileum to rectum. [91]
Cloacal dysgenesis sequence (CDS)
Cloacal dysgenesis sequence is seen in 1:50000 to 250,000 neonates and presents with a phallus-like structure, smooth perineum, and absence of urethral, vaginal, and anal openings. [92] Ultrasound findings include: echogenic bowel, dilated colon, perineal mass, dilated bladder, and fetal hydrops. [38] Infants with cloacal dysgenesis sequence (CDS) have no egress for urine and usually have renal failure and pulmonary hypoplasia, but survival has been reported. [93]
Cloacal malformation
Cloacal malformations are characterized by a communication between the gastrointestinal, urinary, and genital structures resulting in a single opening on the perineum. [94] Ultrasound findings include massive polyhydramnios and dilatation of the bowel. [95] Cloacal atresia may also exist with multiple atresias of the small bowel and the colon, representing a possible new autosomal recessive defect. [41]
Cystinuria
A hyper-echogenic colon [96] and a tubular echogenic mass noted at < 36 weeks and filling contiguous portions of the fetal colon have been associated with cystinuria. [97] The antenatal biochemical expression of cystinuria and its relationship to fetal hyper-echogenic colon is discussed elsewhere. [98] Finally, an echogenic colon may also reflect the presence of an entero-urinary fistula. [99]
Treatment
A future treatment option may be gene delivery to the fetal gut by ultrasound guided gastric injection, which could ultimately prevent the early-onset of intestinal diseases such as cystic fibrosis. [100]
Sacrococcygeal Tumor (SCT): Information
Page Links: Definition, Prevalence, Classification
Definition
Sacrococcygeal tumors (SCT) are among a varied group of fetal neoplasms. SCTs are composed of ectoderm, mesoderm, and endoderm. The tumor likely develops during the embryonic period with the failure of the primitive streak to differentiate normally into three cell layers. SCTs represent the most common histologic type and the most common fetal neoplasm. Neuroblastomas are the most common type of malignancies. [101]
Prevalence
The prevalence of SCT is one in 20,000 to 40,000 births. The female to male ratio is about 3:1. [102]
Classification
A classification scheme was devised by Altman and the American Academy of Pediatrics [103]:
Type 1: Tumors predominantly externally attached to the coccyx that may have a small presacral component.
Type 2: Tumors have both an external mass and significant pelvic extension.
Type 3: Tumors are visible externally, but the predominant mass is pelvic and intra-abdominal.
Type 4: Tumors are not visible externally, but are entirely presacral.
While the classification scheme was devised for neonates, inferences can be made for prenatal ultrasound.
The major risk from SCT is rapid tumor growth. Large tumors result in hemodynamic complications which can result in fetal hydrops. This complication largely defines outcome. [104]
Malignancy is always a possibility with SCTs and optimal management includes delivery in a tertiary care setting. [105] There is a high association between fetal hydrops and death. [106] During the development of fetal hydrops, umbilical vein pulsations and increased aortic blood flows are observed. [107]
Cardiovascular Changes
Fetal hydrops is a result of high output cardiac failure caused by increased tumor vascularity. The increase in blood flow through the SCT acts as an AV fistula, which causes high output cardiac failure. [108]

Fetuses with high output failure from SCTs demonstrate the following on 2-D Doppler echocardiography: dilated ventricles, pericardial effusion, increased velocities and volume flows, tricuspid regurgitation, and mitral regurgitation. [8]
In addition, high output failure is associated with placental enlargement, increased flow through the SCT, and enlargement of the inferior vena cava. [109] Dilated ventricles maintain a normal fractional shortening index, while fetal hydrops is manifest by pleural effusion as well as pericardial effusion. [110] Combined ventricular output is high, the descending aortic flow and placental flow are increased, and high velocity arterial flow is seen in the tumor. [10]
Prognostic Factors
Prognostic factors associated with high output failure include cardiomyopathy and increased preload indices of the fetal venous system. [111] Tumor composition predicts tumor behavior. Solid SCTs are more likely to be highly vascularized and are associated with fetal hydrops. Cstic SCTs are more likely to be associated with a non-hydropic fetus. [112]
Larger tumors have a poor prognosis. Tumor volume to fetal weight ratio is predictive of outcome for SCT fetuses at < 24 weeks gestation. [113]
Solid tumor volume to head volume ratio is also predictive of fetuses with high risk for poor outcome. [114] Other poor prognostic factors include tumor size that is greater than the biparietal diameter, increased placental size, and polyhydramnios. [115] Other outcome predictors include diagnosis at an early gestational age and premature delivery. [116]
Umbilical artery Doppler studies are relevant for prognosis. Sudden fetal demise in the absence of fetal hydrops is seen in SCT fetuses with reversed end diastolic umbilical arterial flow. [117]
Fetal Surgery
Since large fetal tumors can result in fetal death from anemia and/or high output cardiac failure, fetal surgery and resection of the mass is a potential treatment option. [118],[119] Membrane separation is a possible complication of fetal surgery. However, two reported open fetal surgical cases for SCT were successfully managed without chorioamnionitic membrane separation. [120] There are other reported successful outcomes for prenatal surgical interventions in cases of SCT. [121],[122] While fetal surgical resection may be an option, preterm labor continues to be a potential complication. [123] Evaluative methods to explore the feasibility of fetal surgery include accurate ultrasound, MRI, and fetal echocardiography. [124] As selection criteria improve, fetal surgery may correspondingly improve outcomes. [125] Ultrasound evaluative methods remain critical to operative success. [126]
During fetal surgery, significant cardiovascular changes occur that amplify the already high physiological output present. [127] Increased preload and afterload can be defined with echocardiography. [128]
Trends in operative intervention for SCTs include minimally invasive techniques such as fetoscopic resection and the use of radio frequency ablation (RFA). [129],[130] However, percutaneous laser ablation of SCT tumors may result in necrosis and hemorrhage within the mass leading to fetal death. [131]
Treatment using Radio Frequency Ablation
Radiofrequency ablation of the SCT may result in fetal death due to hemorrhage into the tumor, while targeted ablation of feeding vessels appears to increase the chances for survival. [132] Complications of intrauterine RFA include hip dislocation and sciatic nerve injury. [133] Use of an insulated wire for thermal coagulation of fetal vessels has not met with success. [134]
Treatment using Prenatal Drainage
Drainage of a cystic SCT is possible. Approximately 15% of SCTs are cystic, and can result in intra-pelvic obstruction of the urinary tract. [135] Prenatal decompression can alleviate the obstructive effects. [136],[137]
Neonatal Operative Management
Surgical approaches for neonatal operative management of SCTs diagnosed in utero include consideration of the following: early resection, abdomino-perineal approach, supine position, and devascularization of the tumor. [138]
Some reports suggest survival as early as 27 weeks following neonatal resection. [139] Type 1 tumors (solid and cystic elements with no pelvic extension) have been successfully resected at 34 weeks gestation after neonatal stabilization. [140] Occasional malignant transformation of a SCT has been noted as long as 11 months following initial resection. [141] Some SCTs demonstrate organ differentiation indicating, for example, the presence of small bowel within the tumor. [142] In other instances, it may be difficult to distinguish a differentiated teratoma from a fetus-in-fetu. [143],[144] Histologic changes occur in SCTs over time with transitions from malignant elements to nonmalignant elements and/or other findings of tumor maturation. [145]
Neonatal Outcome
In a study of 21 fetuses with a SCT diagnosed by ultrasound, the in utero mortality rate was 19% and the incidence of premature labor was 50%. [146] In another series of 30 patients [147], gestational age at diagnosis was 23.9 weeks, while 14 of 30 fetuses survived. Of 26 continuing pregnancies, 81% had complications. Four of 30 underwent fetal resection and three of four fetuses survived. [47]
In 41 patients with a SCT diagnosed prenatally, 12 underwent various fetal interventions. Six of 12 had long-term survival (50%). [148] Seventeen fetuses had no intervention, were born at a median of 38 weeks gestation, and postnatal surgical excision resulted in good outcomes. The overall survival rate for SCT was 77%. [48] Others report a perinatal mortality rate of 62.5% in fetuses with a SCT. All deaths occurred in premature infants. [149]
In summary, outcomes improve when there are efforts to control the neonatal hyperdynamic state with initial devascularization procedures followed by staged resection. [150] To obtain optimum outcome, serial cardiac surveillance, and assessment of the size of the SCT are important since size determines obstetrical risks and mode of delivery. Tumors which grow at > 150 cm per week are at increased risk for poor outcome. [151] Associated anomalies affect outcome. “Fetal hydrops, orthopedic impairment such as lower extremity weakness and swelling, and urinary incontinence are important clinical factors affecting the outcome after birth in prenatally diagnosed SCT.” [152] In Type III SCT (tumor extends to the pelvis), patients may present with a variety of related abnormalities including hydronephrosis, neurological deficits such as paralytic talipes equines, and bladder or bowel dysfunction. [52]
Spontaneous rupture of a SCT can occur in utero with consequent fetal anemia. [153],[154] Prevention of spontaneous rupture during labor is possible with appropriate antenatal surveillance.
Neonatology Perspective
Total removal of the SCT is considered curative. These patients are followed with serial tumor markers (beta HCG, alpha FP, and alkaline phosphatase levels) to determine whether resection was complete.
Sacrococcygeal Tumor (SCT): Imaging Considerations
Sacrococcygeal teratomas (SCT) are fetal tumors with malignant potential. [155]
Sacrococcygeal teratomas represent the most common neoplasms in the fetus and newborn. Prenatal ultrasound can facilitate a multidisciplinary approach to care. [156] Prental diagnosis is important because fetal SCTs may behave differently compared to neonatal SCTs. [157] Prenatal diagnosis can also help to determine optimal delivery time and delivery environment. [158] In addition to ultrasound diagnosis, alpha fetal protein may be elevated or normal in maternal serum, and acetyl cholinesterase may be present in amniotic fluid. [159]
2-D Ultrasound Diagnosis
The most common findings on prenatal ultrasound include a mass and polyhydramnios. [160] Sacrococcygeal teratoma may be diagnosed during the first trimester by utilizing either 2-D or 3-D ultrasound. [161],[162] Ultrasound characteristics of sacral sacrococcygeal teratomas include a large, predominantly solid mass with scattered cystic elements in the presence of polyhydramnios. [163]
Color Doppler is useful and can differentiate between a cystic sacrococcygeal teratoma and a meningomyelocele. [164]
The following are ultrasound criteria: 1. heterogeneous mass, 2. fetal hydrops, 3. polyhydramnios, and 4. tumor weight 50% above that of the fetal weight. [165] A rapid growth or increase in the vascular bed may lead to fetal anemia and hypo-proteinemia which are poor prognostic factors. [11]
Three sonographic patterns are: 1. Mixture of solid and cystic components, 2. predominantly solid with scattered anechoic areas and, 3. unilocular cystic mass. [166] Germ cell tumors exhibit ossification and calcifications are present in a significant number of cases. Fetal hydrops is an associated finding and represents a late sign of cardiac failure. [167]
Ultrasound can distinguish internal and external locations of the SCTs as well as predominantly cystic and mixed tumors. [168] Location may be entirely external or predominantly external with pelvic extension. [169]
3-D Ultrasound
Three-dimensional ultrasound helps to determine tumor size and location [170], and the presence and the location of fetal vessels. [171] 3-D ultrasound allows precise information of the fetal sacral and pelvic area [172], and may distinguish between a cystic SCT and a meningocele. [173]
MRI
MRI is useful in defining intra-pelvic extension of SCTs, [174] and ultra-fast T 2 weighted imaging permits morphologic details of the tumor. [175] Other attributes of MRI include: large field of view, excellent soft tissue contrasts, and accurate volume measurements. [176] In a series of fetal tumors assessed with MRI, information provided by MRI enhanced perinatal care. [177] While there is general agreement between MRI and ultrasound, MRI information more accurately defines extension of the fetal tumor. [178]


Above. Case 1. 24 3/7 weeks gestation. Sacrococcygeal teratoma. Longitudinal scan of lower spine showing sacrum with associated mixed cystic and solid mass.
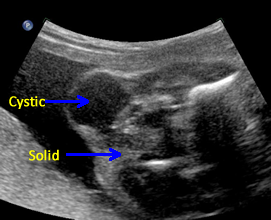
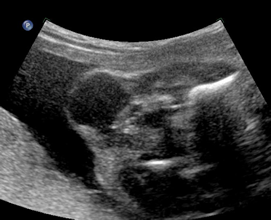
Above. Case 1. 24 3/7 weeks gestation. Sacrococcygeal teratoma. An oblique scan through the same area shows both solid and cystic areas as well as calcifications, which are commonly seen in teratomas. The mass appeared completely external.
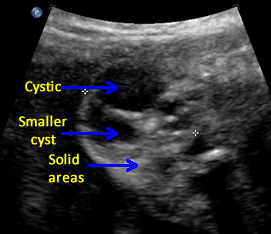
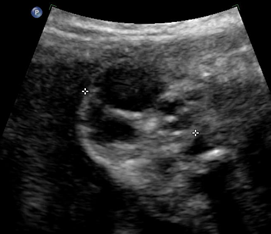
Above. Case 1. 24 3/7 weeks gestation. Sacrococcygeal teratoma. Similar part of the tumor as above showing cysts of various sizes.
Above. Case 1. 24 3/7 weeks gestation. Sacrococcygeal teratoma. The predominant cyst is large and contains anechoic fluid. No polyhydramnios or evidence for fetal hydrops were noted. Delivery occurred at term. Uneventful resection and repair occurred on day 2 of life.


Above. Case 2. 32 1/7 weeks gestation. Sacrococcygeal teratoma. Coronal view of pelvic area. External mass in low sacral area with both cystic and solid components, and little blood flow. The mass appeared completely external to the fetal abdomen.
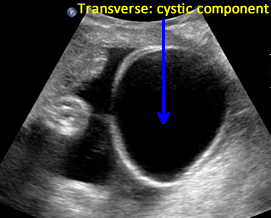

Case 2. 32 1/7 weeks gestation. Sacrococcygeal teratoma. Some transverse views through the predominant cyst demonstrate no solid areas.
Above. Case 2. 32 1/7 weeks gestation. Sacrococcygeal teratoma. Some views demonstrate both solid and cystic components. There was no evidence for fetal hydrops or polyhydramnios. Delivery occurred following premature labor at 34 1/7 weeks. Skin completely covered the defect and an uneventful repair occurred.
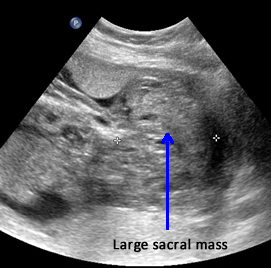
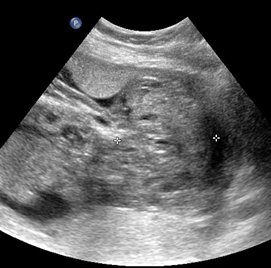
Above. Case 3. 25 4/7 weeks gestation. Sacrococcygeal teratoma. Large mass measuring 5.9 x 10.9 x 8.3 cm, which is noted in the sacral area, but also extends internally superior to the bladder.


Above. Case 3. 25 4/7 weeks gestation. Sacrococcygeal teratoma. Color Doppler indicates the vascular nature of the external portion of this tumor.


Above. Case 3. 25 4/7 weeks gestation. Sacrococcygeal teratoma. Coronal scan demonstrates internal extension of the tumor to the lower abdomen superior to the bladder and within the bounds of the 2 umbilical arteries. This tumor represents a Type 2 sacrococcygeal teratoma (extension to the abdomen).

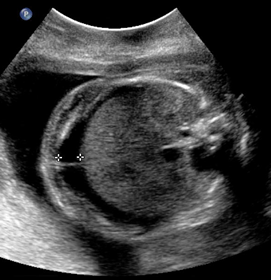
Above. Case 3. 25 4/7 weeks gestation. Sacrococcygeal teratoma. Transverse of the abdomen demonstrates fetal ascites and likely fetal hydrops.


Above. Case 3. 25 4/7 weeks gestation. Sacrococcygeal teratoma. Other signs of fetal hydrops include scalp edema.


Above. Case 3. 25 4/7 weeks gestation. Sacrococcygeal teratoma. Cardiomegaly is apparent with heart filling most of the chest cavity.


Above. Case 3. 25 4/7 weeks gestation. Sacrococcygeal teratoma. Peak systolic velocity of the middle cerebral artery demonstrates fetal anemia. The patient delivered prematurely and early neonatal death occurred as a result of multi-organ failure.
Sacrococcgeal teratoma (SCT)
Above. Large sacrococcgeal teratoma (SCT) with mixed solid and cystic elements.
Above. Sacrococcgeal teratoma. This tumor has a vascular component creating a risk for arteriovenous shunt, fetal anemia, and resultant fetal hydrops.
Above. Sacrococcgeal teratoma. Same patient as above demonstrating fetal hydrops. Note Ascites (A), Liver (L), and Bowel (B).
Above. Note cardiac enlargement with heart filling the predominant portion of the fetal chest.
Above. Sacrococcgeal teratoma, Type 1 (No tumor extension to the abdomen). This is an example of a predominantly cystic type.
Above. Sacrococcgeal teratoma. Same patient as immediately above. No fetal hydrops.
-
Abstract: PMID: 17436317 -
Abstract: PMID: 17652931 -
Abstract: PMID: 16764013 Ahmadi F, Vosough Taghi Dizaj A, Irani S. Prenatal diagnosis of fetal umbilical vein varix in an intracytoplasmic sperm injection conception: a case report. Iran. J. Radiol. 2007;4(2):117-119. -
Abstract: PMID: 16389043 -
Abstract: PMID: 15042382 -
Abstract: PMID: 16498367 -
Abstract: PMID: 15685644 -
Abstract: PMID: 11211133 -
Abstract: PMID: 15305101 -
Abstract: PMID: 11169296 Cruise K, Rouse G. Klippel-Trenaunay-Weber Syndrome complicated by extra-fetal umbilical vein varix. Journal of Diagnostic Medical Sonography. 2002;18(5): 317-320. -
Abstract: PMID: 10864059 -
Abstract: PMID: 16749967 -
Abstract: PMID: 11169334 -
Abstract: PMID: 15229845 -
Abstract: PMID: 3898782 -
Abstract: PMID: 17654754 -
Abstract: PMID: 7472957 -
Abstract: PMID: 19453080 -
Abstract: PMID: 6351003 -
Abstract: PMID: 7099748 -
Abstract: PMID: 9174025 -
Abstract: PMID: 3418475 -
Abstract: PMID: 18405693 -
Abstract: PMID: 19319541 -
Abstract: PMID: 16888707 -
Abstract: PMID: 1200245 -
Abstract: PMID: 710040 -
Abstract: PMID: 622496 -
Abstract: PMID: 10813334 -
Abstract: PMID: 11685446 -
Abstract: PMID: 572092 -
Abstract: PMID: 7355213 -
Abstract: PMID: 10663833 Link: http://en.wikipedia.org/wiki/Meconium_peritonitis -
Abstract: PMID: 14634976 -
Abstract: PMID: 10955566 -
Abstract: PMID: 12889504 -
Abstract: PMID: 16049997 -
Abstract: PMID: 19126503 -
Abstract: PMID: 12835586 -
Abstract: PMID: 10663833 -
Abstract: PMID: 12497690 -
Abstract: PMID: 10203675 -
Abstract: PMID: 17465298 -
Abstract: PMID: 12192485 -
Abstract: PMID: 11127113 -
Abstract: PMID: 17206405 -
Abstract: PMID: 17036237 -
Abstract: PMID: 15136891 -
Abstract: PMID: 18648207 -
Abstract: PMID: 11536273 Iwabuchi Toshihisa, Shimotake Takashi, Tomiyama Hideki, Fumino Shigehisa, Iwai Naomi. A case of giant cystic meconium peritonitis (GCMP) treated by peritoneal drainage and radical operation in the neonatal period. Journal of the Japanese Society of Pediatric Surgeons. 2002;38(4):702-706. -
Abstract: PMID: 3546721 -
Abstract: PMID: 12601839 -
Abstract: PMID: 2051534 -
Abstract: PMID: 18838290 -
Abstract: PMID: 21193703 -
Abstract: PMID: 15685665 -
Abstract: PMID: 11982991 -
Abstract: PMID: 16757916 -
Abstract: PMID: 3300224 -
Abstract: PMID: 1552452 -
Abstract: PMID: 1442962 -
Abstract: PMID: 8469599 -
Abstract: PMID: 16147839 -
Abstract: PMID: 15662698 -
Abstract: PMID: 10423811 -
Abstract: PMID: 7899286 -
Abstract: PMID: 1336928 -
Abstract: PMID: 11484210 -
Abstract: PMID: 2661823 -
Abstract: PMID: 12385942 -
Abstract: PMID: 3530266 -
Abstract: PMID: 9572387 -
Abstract: PMID: 16888705 -
Abstract: PMID: 16731893 -
Abstract: PMID: 18577677 -
Abstract: PMID: 18773674 -
Abstract: PMID: 12874733 -
Abstract: PMID: 12837448 -
Abstract: PMID: 22362191 -
Abstract: PMID: 20069658 -
Abstract: PMID: 21199752 -
Abstract: PMID: 15645250 -
Abstract: PMID: 12738110 -
Abstract: PMID: 15065109 -
Abstract: PMID: 16491007 -
Abstract: PMID: 19735835 -
Abstract: PMID: 19921644 -
Abstract: PMID: 18164419 -
Abstract: PMID: 20006002 -
Abstract: PMID: 12862274 -
Abstract: PMID: 19938077 -
Abstract: PMID: 21270932 -
Abstract: PMID: 21225668 -
Abstract: PMID: 17202504 -
Abstract: PMID: 16888707 -
Abstract: PMID: 16839275 -
Abstract: PMID: 15653597 -
Abstract: PMID: 20088402 -
Abstract: PMID: 4843993 -
Abstract: PMID: 21656530 -
Abstract: PMID: 12013082 -
Abstract: PMID: 10362082 -
Abstract: PMID: 9931825 -
Abstract: PMID: 3287252 -
Abstract: PMID: 2286911 -
Abstract: PMID: 2656957 -
Abstract: PMID: 15192290 -
Abstract: PMID: 10693688 -
Abstract: PMID: 21683219 -
Abstract: PMID: 19752518 -
Abstract: PMID: 8327820 -
Abstract: PMID: 9788651 -
Abstract: PMID: 14646415 -
Abstract: PMID: 2658603 -
Abstract: PMID: 8062028 -
Abstract: PMID: 12913340 -
Abstract: PMID: 12557042 -
Abstract: PMID: 21092543 -
Abstract: PMID: 10630541 -
Abstract: PMID: 11914697 -
Abstract: PMID: 11754151 -
Abstract: PMID: 12418767 -
Abstract: PMID: 15353490 -
Abstract: PMID: 15360113 -
Abstract: PMID: 14533891 -
Abstract: PMID: 19260783 -
Abstract: PMID: 19129708 -
Abstract: PMID: 11228510 -
Abstract: PMID: 12596115 -
Abstract: PMID: 11857611 -
Abstract: PMID: 9813425 -
Abstract: PMID: 11746122 -
Abstract: PMID: 10442611 -
Abstract: PMID: 11283874 -
Abstract: PMID: 18675620 -
Abstract: PMID: 18581776 -
Abstract: PMID: 17561476 -
Abstract: PMID: 14507007 -
Abstract: PMID: 17183856 -
Abstract: PMID: 9589148 -
Abstract: PMID: 9498382 -
Abstract: PMID: 9660225 -
Abstract: PMID: 15017565 -
Abstract: PMID: 16481257 -
Abstract: PMID: 10701852 -
Abstract: PMID: 7738756 -
Abstract: PMID: 19122459 -
Abstract: PMID: 18937757 -
Abstract: PMID: 16958151 -
Abstract: PMID: 16912491 -
Abstract: PMID: 3548370 -
Abstract: PMID: 3903582 -
Abstract: PMID: 3332744 -
Abstract: PMID: 2681672 -
Abstract: PMID: 3332743 -
Abstract: PMID: 15213888 -
Abstract: PMID: 21387328 -
Abstract: PMID: 15170807 -
Abstract: PMID: 10636053 -
Abstract: PMID: 9259890 -
Abstract: PMID: 1955658 -
Abstract: PMID: 3047782 -
Abstract: PMID: 2310588 -
Abstract: PMID: 9239831 -
Abstract: PMID: 10687774 -
Abstract: PMID: 19642602 -
Abstract: PMID: 16389043 -
Abstract: PMID: 11876816 -
Abstract: PMID: 19484739 -
Abstract: PMID: 9435729 -
Abstract: PMID: 10405065 -
Abstract: PMID: 11215715 -
Abstract: PMID: 21316891 -
Abstract: PMID: 11756117
4
12
36
54