Spina Bifida (Neural Tube Defects)

Page Links on Spina Bifida Topics: Definition, Prevalence, Classification, Risk Factors, Etiology, Genetics, Prevention, Outcome, Differential Diagnosis, References
Definition of Spina Bifida
The following definitions are from National Library of Medicine – Medical Subject Headings, 2008 MeSH.
Meningocele: A congenital or acquired protrusion of the meninges, unaccompanied by neural tissue, through a bony defect in the skull or vertebral column.
Meningomyelocele: Congenital, or rarely acquired, herniation of meningeal and spinal cord tissue through a bony defect in the vertebral column.
Spinal dysraphism: Congenital defects of closure of one or more vertebral arches, which may be associated with malformations of the spinal cord, nerve roots, congenital fibrous bands, lipomas, and congenital cysts. These malformations range from mild (spina bifida occulta) to severe, including rachischisis where there is complete failure of neural tube and spinal cord fusion, resulting in exposure of the spinal cord at the surface. Spinal dysraphism includes all forms of spina bifida. The open form is called spina bifida cystica and the closed form is spina bifida occulta.
Fetal Prevalence of Spina Bifida

Above. United States data show the prevalence of neural tube defects has declined due to: preconceptual supplementation with folic acid and, prenatal diagnoses with resultant selective termination of pregnancies.

Above. There is significant geographic variations in the rates of spina bifida and anencephaly. The rates (per 10,000 population) for neural tube defects are twice in certain areas of Russia compared to Norway, for example. [1],[2],[3],[4],[5],[6],[7]
*Sweden, 6.1 = 2.4/10,000 newborns plus 3.7/10,000 termination of pregnancy.


In summary, the incidence of neural tube defect is affected by racial, geographic and seasonal variations as well as the risk factors listed above. Most NTDs which are not related to a syndrome are multifactorial in origin.
There is also an increased frequency in: maternal hyperthermia valproate ingestion.
No Mendelian inheritance pattern is noted.
Genetic contribution evidence is present due to the following: single gene defects, recurrence among siblings, and higher likelihood in twins.
Classification
Neural tube defects can be classified as either abnormalities of the skull or brain. Those of the skull and brain include Arnold Chiari malformation, which represents secondary changes within the fetal brain associated with spinal defects. [8]
A separate classification includes abnormalities of the vertebral arches (dysraphism) and includes open defects such as meningocele and spina bifida cystica. [8] Spina bifida occulta represents a closed spinal defect. [9]



Above. A separate classification for Spinal Dysraphism (spinal defects which affect the vertebra arches) has been proposed. [10] This classification divides the spinal lesions into open and closed defects and suggests certain associations with each.
Risk Factors

Above. Neural tube defects can broadly be described as syndromic and non-syndromic. [11] In the non-syndromic multifactorial group, a number of specific risk factors have been reported.

Above are risk factors reported for spina bifida. [11]
Exposure to agricultural pesticides and to fumonisin, a mycotoxin that often contaminates corn, appear as causative links for the occurrence of neural tube defects. [12],[13]
Retinoid acid (RA) plays a role in the development of the spinal cord while genetic polymorphism of the human retinoic acid-metabolizing enzyme is being explored for its role in the development of spina bifida.
Etiology

Above. The etiology of neural tube defects is related to environmental and genetic factors and may be considered heterogeneous and multifactorial. The genetic component is discussed separately.

Congenital spinal defects are associated with intracranial changes and the Arnold Chiari malformation. The Chiari malformation, associated with hindbrain herniation, is related to low spinal pressure relative to cranial pressure and results in hindbrain-related symptoms caused by cerebellar and brainstem compression. [16] Increased CNS volume, with any space-occupying lesion, compresses veins and raises venous pressure which in turn raises the volume of cerebrospinal fluid (CSF). When blood enters the CNS during fetal movement, the compressed posterior fossa prevents the spine-to-head flow of CSF. These movement-induced pressure events may result in spinal cord injury.
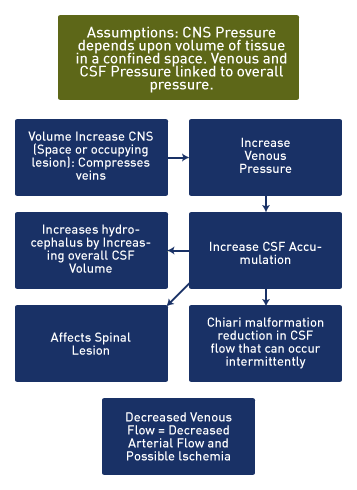
Above. Williams [17] proposes a venous hypothesis for spinal lesions and hydrocephalus related to the overall pressure in the CNS, which is dependent upon all of the volume within the closed CNS space. Arterial supply is maintained normally because of venous outflow from the CNS. Increased CNS volume with any space-occupying lesion compresses veins and raises venous pressure, which in turn raises the volume of cerebrospinal fluid (CSF). These changes affect hydrocephalus and the associated spinal lesions, while the Chiari malformation leads to reductions in CSF flow that can occur intermittently.
Genetics

Above. Neural tube defects are due to multiple environmental and genetic factors. [18]

Above. These gene-gene factors and gene-environmental factors are operable through a variety of maternal conditions. [23]

Above. With respect to genetic risk factors, multiple genes are responsible.

Above. Among fetuses with open neural tube defects, a rate of chromosomal abnormalities of 7% has been reported. [19] Among 10 fetuses: 3/21 (14%) were encephalocele defects, 6/66 (9%) were spina bifida abnormalities and 1/57 (2%) were lethal defects. [24]

Above. Specific chromosomal abnormalities have been reported with neural tube defects and are variable [20], while the risk factors for a chromosomal abnormality vary with:
maternal age,
gestational age at diagnosis,
the presence of other malformations family history of chromosomal abnormalities.
In 587 women, when central nervous system malformations were isolated, specific associations were noted with Trisomy 21, Trisomy 13, and Trisomy 18. [21]

Above. Both chromosomal structural deletions and duplications have been associated with NTDs. [22]

Above. Over a 6-year period, 144 fetuses with open NTD underwent prenatal chromosome analysis between 12 and 37 weeks of gestation as part of a prospective, multicenter prenatal diagnosis and counseling program. The prevalence of chromosomal abnormality varied with the defect, with a 14% (3/21) prevalence among those with cephalocele, a 9% prevalence (6/66) among those with spina bifida, and a 2% (1/57) prevalence among those with lethal defects such as acrania, anencephaly or iniencephaly. [24]

Above. Genetically detectable human chromosome abnormalities were studied to identify patients with 14 brain malformations including 542 with deletions, and 290 carrying duplications. [23] Among CNS malformations many chromosomal deletions and duplications were identified among associated bands in various regions.

Many folate metabolism-related genes have been reported. [24] The gene VANGL-1 and its mutations have been implicated as risk factors for the development of neural tube defects. [25] Other specific VANGL-1 mutations have been reported. [26] The endothelial nitric oxide synthase gene has been implicated as a risk factor in women with impaired homocysteine metabolism. [27] Some deletion allele may be a protective NTD genetic factor [28] but these effects remain under investigation. [29] Specific maternal genotypes such as MTRR 66GG genotype are a maternal risk factor for spina bifida especially when intracellular vitamin B12 status is low. [30]
Prevention of Spina Bifida

Above. In the United States, mandatory folic acid fortification has resulted in a significant decline in spina bifida.
United States data. Change in Spina bifida prevalence among all participating systems. Prevalence per 10,000. [31]
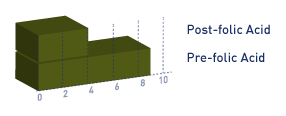
Above. Note the pre-and post-Folic acid fortification in Canada.[32]

Above. Folic acid fortification programs demonstrate a positive but variable influence on the prevention of defects.
The outcome of 33 Infants Studied for Spina Bifida

Above. In an effort to determine whether prenatal ultrasound findings in fetuses with open spinal defects predict ambulatory status after birth, 33 infants were studied. [33] All infants with L4 lesions and below were ambulatory while no infants with thoracic lesions were ambulatory.
The site of the spinal lesion is the most significant predictor of intellectual outcome followed by the presence and degree of ventriculomegaly. The presence of a foot abnormality had no significant effect on the outcome while high spinal lesions and the degree of ventriculomegaly correlated with adverse outcome measures. [34]

Muscle echogenicity is increased in fetuses with open spina bifida. [35] In utero leg movements are present only to disappear within one week after birth.[40] The initial presence of neonatal leg movements does not indicate functional lower motor neuron innervations. [36] Additional neuromuscular damage after the prenatal period may account for the neonatal loss of leg movements. [37]
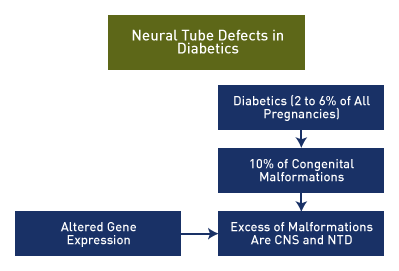
Above. Diabetics comprise 2% to 6% of all pregnancies. Overall 10% of Diabetics have some form of fetal malformation involving a variety of organ systems, while NTDs are commonly represented among CNS malformations. [38]

Above. A number of mechanisms may be responsible for the increased risk of CNS birth defects among women with diabetes and maternal obesity. [43],[39],[40],[41],[42]

Above. The likelihood for a neural tube defect increases with the degree of maternal obesity, [43] and through a shared mechanism, maternal obesity and gestational diabetes may increase the risk for central nervous system birth defects. [44]

Above. In women with epilepsy, congenital malformations are increased. The highest relative risk for malformations in the offspring of women with epilepsy was observed in spina bifida. [45] In spina bifida patients, epilepsy correlates with coexisting cerebral abnormalities and appears to be multfactorial in origin. [46],[47]
Page Links: Imaging Considerations, References
Imaging Considerations
Ultrasound versus Alpha-Fetoprotein
Most studies suggest routine ultrasound is superior to AFP as a screening test alone for neural tube defects and is likely to replace routine AFP surveillance. [48],[49],[50],[51]
AFP sensitivity for the diagnosis of NTDs can be improved by using ultrasound to confirm dates to reduce gestational age error in the calculation of the AFP result. [3]
Multi-planar views of CNS anatomy offer excellent visualization and improved diagnostic accuracy. [52]
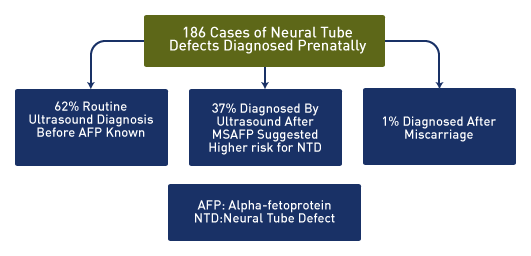
Above. In 189 patients with neural tube defects, 102 underwent MSAFP screening. The results of MSAFP testing were negative in 25 (25%). Without other testing, these 25 NTD patients would have been missed. These included 15 with spina bifida, 6 with encephalocele and 4 with anencephaly. [53]
Overall, competent ultrasound screening replaces amniotic fluid AFP and cholinesterase measurements for the diagnosis of NTDs. [54]
Ultrasound Diagnosis
The diagnosis of open spinal defects has been reported as early as the first trimester using trans-vaginal ultrasound. Findings include a step or angulations of the posterior aspect of the embryo. Abnormal contours of the head and spine as well as empty or enlarged intracranial contents may be seen.
The findings related to an open spinal defect are commonly associated with alterations of the brain anatomy. One of the most common of these is the Arnold Chiari, or Chiari II malformation. The cerebellum is displaced inferiorly due to the open spinal defect which results in ultrasound changes which can be noted before birth.
Above. The ultrasound detection of neural tube defects is accompanied by intracranial signs. The skull itself is commonly “lemon” shaped. Due to the Arnold Chiari II malformation, the cerebellum is effaced creating the “banana” sign and the posterior fossa is frequently obliterated. The dilatation of the lateral ventricles completes the major intracranial signs of open spinal defects. [55],[56]
Above. The axial view of the head to obtain the biparietal diameter will demonstrate dilatation of the lateral ventricles and the “lemon” shaped configuration of the fetal head suggesting a neural tube defect. The cerebellar view will demonstrate effacement of the cerebellum and obliteration of the posterior fossa.
Above. The “lemon” shaped configuration of the fetal head is associated with certain open neural tube defects.
Above. “Banana” shaped cerebellum (yellow arrow) and obliteration of posterior fossa (green arrow) is also associated with certain open neural tube defects.
Above. In most large ultrasound series, open spinal lesions were associated with classical findings of the banana sign or lemon sign while the closed lesions did not present with intracranial changes. [57]
Above. Ultrasound signs were studied for the diagnosis of spina bifida during mid-trimester scans performed between 18 and 28 weeks; the lemon sign was noted in over 50% of cases while small cerebellum, effaced cisterna magna, small posterior fossa and visualization of spinal defect were present in over 90% of cases. [58] Ventriculomegaly was noted in 81% and the spinal defect was observed in 98%. [11]
Above. Enlargement of the ventricles or ventriculomegaly is seen with open spinal lesions and the associated Chiari II malformation. The other major causes of ventriculomegaly include aqueductal stenosis, Dandy-Walker complex and absence of the corpus callosum. [59],[60],[61]
Above. Supratentorial abnormalities in the Chiari II malformation are reported in a series of papers. [62],[63],[64] The shape of the occipital horn of the lateral ventricle is described as pointed rather than rounded; the tectum is described as elongated or beaking while an interhemispheric cyst is sometimes present. Others suggest that occipital pointing was present in up to 77.3% of fetuses with spina bifida. [65]
Above. Views of the posterior horn of the lateral ventricle can suggest a triangle sign, which is associated with a low vertebral open spinal lesion, while a square appearance of the posterior horn of the lateral ventricle on coronal views suggests an upper spinal lesion. [66]
Associated Malformations
Above. In this study, associated malformations are described in infants with neural tube defects. 360 infants were born with neural tube defects among 334,262 consecutive births. Associated malformations were more frequent in infants with encephalocele (37.5 %) than in infants with anencephaly (11.8 %) or infants with spina bifida (23.7 %). [67]
Above. In 51 cases of central nervous system defects, 24 cases were spina bifida and 27 cases were anencephaly, exencephaly and encephaloceles. Among these 51 cases, other associated anomalies included: isolated CNS, other neural defects, midline or lateralization anomalies and musculoskeletal, urogenital, vascular anomalies as well as known syndromes. [68]
In the above series, 78 of 178 patients presented with associated anomalies. 50% of the anomalies came from fetuses with sacral or lumbosacral lesions. In those 100 with spina bifida only, 88% had sacral or lumbosacral lesions. [69] Tomographic ultrasonographic techniques may further increase diagnostic accuracy.[70]
Accuracy
Above. In the European Multi-Center Study, of the 252 cases of isolated spina bifida, 171 (68%) were correctly identified prenatally; 66% of these before 24 weeks’ gestation. The diagnosis was missed on scan in 24% cases and 8% were not scanned. [71]
Above. Antenatal definition of the level of the spinal lesion may be difficult with standard ultrasound application. The accuracy of defining the upper level of the spina bifida defect suggested that community physicians were less likely to define the accurate level of the lesion compared to academic staff physicians. Neither group agreed exactly with the the level of the lesion compared to the actual level determined after birth although academic physicians agreed within 1 level of the lesion 78% of the time. [72] However, ultrasound detection rates are variable and reported as low as 24% in some populations. [73]
Above. A postmortem evaluation was made of 220 prenatally diagnosed fetuses with neural tube defects. Additional malformations not detected by prenatal ultrasound were noted on postmortem examination in 21.6% of the cases. [74]
MRI and Ultrasound in Diagnosis of Neural Tube Defects
Ultrasound plays an important role in the detection of neural tube defects while MRI may be especially useful in the evaluation of specific abnormalities. [75] Advances in MRI technology include rapid pulse sequences, parallel imaging and improved coil design. Recent applications include in vivo brain development and early diagnosis of congenital malformations and as adjunct to lesions not well visualized with ultrasound. [76],[77] MRI confirms sonographically demonstrated CNS malformations while additional findings may be discovered. In addition, MRI findings may influence parental counseling decisions. [78]
However, some studies demonstrate no significant advantage of antenatal MRI compared to ultrasound in detecting the level of spinal lesions and the presence of ventriculomegaly. [79]
Ultrasound may not define all spinal cord lesions. Diagnostic accuracy is poor in defining some cord abnormalities such as diastematomyelia and segmental spinal dysgenesis but this factor may not make a difference in outcome. [80]
The role of MRI is often considered an accessory imaging modality to confirm questionable lesions or anatomy, to define and to characterize lesions and associated anomalies. [81]
While prenatal MRI readily defines CNS lesions and candidates for neonatal neurosurgery [82], most CNS lesions are appropriately assessed by antenatal ultrasound with the exception of neuronal migration disorders, and ventriculomegaly in the early second trimester of pregnancy. [83], and certain spinal cord anomalies.[33]
In the MRI evaluation of the posterior fossa, the presence of the cerebrospinal fluid signal on on both sagittal and axial images is indicative of a normal posterior fossa while its absence is suggestive of a Chiari 11 malformation. [84]
Spina Bifida Images
The imaging tasks in defining spina bifida are as follows:
I. Assess cranial views in Spina Bifida
A. Shape of cranial bones
B. Arnold Chiari findings
C. Ventriculomegaly
D. Ventricular pointing
II. Assess spinal views in Spina Bifida
A. Understand normal scan planes
B. Determine level of defect
C. Determine type of defect (contents)
D. Determine extent of the defect
E. Measure defect
F. Compare head defect to spine
G. Assess change over time
H. Assess multiple gestations
Cranial Views
Spina Bifida Frontal Bone Scalloping


Above. Fontal bone scalloping and typical “lemon” shaped calvaria represents one of the cranial signs of spina bifida.
Spina Bifida Arnold Chiari Malformation

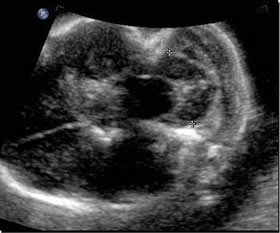




Above. Views using the cerebellar plane demonstrate the Arnold Chiari malformation with short transverse diameter of the cerebellar hemispheres, absence of the posterior fossa (cisterna magna) and effaced or “banana” shaped cerebellum.
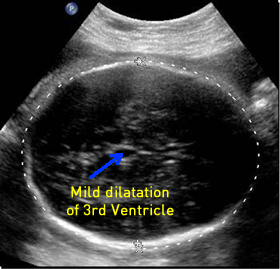
Ventriculomegaly
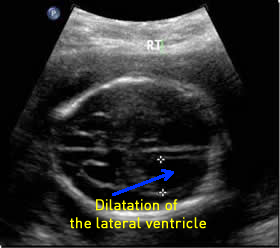

Above. Dilatation of the lateral ventricles is also a potential finding in spina bifida and is often borderline but can be severe and is commonly found at term.
Ventricular Pointing in Spina Bifida


Above. More recent observations suggest a “ventricular point” with the occipital horn of the lateral ventricle pointed rather than rounded. (See Imaging Considerations)
Assess Spinal ViewsUnderstand Normal Scan Planes


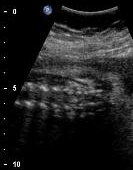
Above. Three normal scan planes for assessing the spine are presented.
Left: longitudinal or sagittal plane; Middle: coronal plane; Right: transverse or axial plane.
Determine Level of the Defect




Above. The last rib is inserted at the level of the 12th thoracic vertebrae. The is the key anatomic site for counting the 5 lumbar vertebrae and for counting the levels of the sacral vertebrae.
Determine the Type of Spina Bifida Defect (contents)








Meningocele: anechoic cystic sac which contains only meninges and CSF (cerebrospinal fluid)
Myelomeningocele: mixed echogenicity with meninges, neural (spinal cord) elements, and CSF.
Myeloschisis: no overlying sac but spinal defect only.
Demonstrate Bony Defects





 Determine Extent of Defect
Determine Extent of Defect

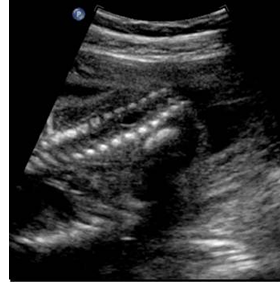


Above. Determining the extent of the lesion is best accomplished by a coronal view of the spine or oblique coronal view.
Using T-12 as a key anatomic site, the lumbar and spinal vertebrae can be numbered.
Measure the Defect: Spina Bifida Image Diagnostics


Above. Measure the defect in the anterior-posterior (AP) and transverse dimensions.
Compare Head to Spine Findings
Patient 1


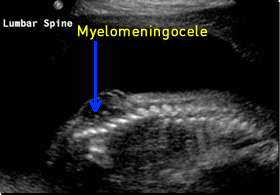

Patient 2



Patient 3
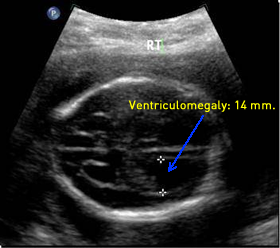


Patient 4



Above. Inspection between head and spine findings is important. Cranial findings may be more severe compared to spine findings, while the reverse is also seen.
Patient 5: Cervical Myelomeningocele


Above. Cervical myelomeningocele (MM). Longitudinal view cervical spine. Fetal head is to the right. Note disruption of the spine.


Above. Cervical MM. Oblique to coronal view. Note level of defect at C5-C6. The view includes the meninges.


Above. Cervical MM. Axial view. The atria of the lateral ventricle is within normal limits at 8.01 mm.


Above. Cervical MM. Cerebellar view. The transverse cerebellar diameter and the posterior fossa measure within normal limits.
Compare Spina Bifida Findings Over Time
Patient A
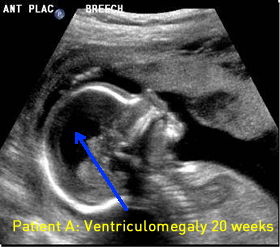






Above. There can be changes in the cranial and spinal findings over time. In this example, both the cerebral and spinal findings showed significant worsening over time.
Assess Spine Differences in Twins


References
Garca Lpez E, Rodrguez Dehli C, Ariza Hevia F, Rodrguez Fernndez A, Fernndez Toral J, Riao Galn I, Mosquera Tenreiro C. Prevalence of neural tube defects in Asturias (Spain): impact of prenatal diagnosis. Gac Sanit. 2009 Nov-Dec;23(6):506-11. Abstract: PMID: 19406531
-
Abstract: PMID: 19353336 Abd El Ghani A, El Ansarry K. Embryological summary. A Neural Tube Defects. [Note: Article defines Spina Bifida, Anencephaly, Encephalocele, and Iniencephaly]. ASJOG. 2006 Feb;3:38-48. Racial/ethnic differences in the birth prevalence of spina bifida: United States, 1995-2005. Centers for Disease Control and Prevention (CDC). MMWR Morb Mortal Wkly Rep. 2009 Jan 9;57(53):1409-13. Abstract: PMID: 19129744
-
Abstract: PMID: 19228313 -
Abstract: PMID: 19412801 -
Abstract: PMID: 16575897 Neural Tube Defects Classification: abnormalities of the skull and brain. National Library of Medicine – Medical Subject Headings, 2008 MeSH, MeSH Descriptor Data Neural Tube Defects Classification: abnormalities of the spine. National Library of Medicine – Medical Subject Headings, 2008 MeSH, MeSH Descriptor Data -
Abstract: PMID: 10952179 -
Abstract: PMID: 18935989 -
Abstract: PMID: 16495467 -
Abstract: PMID: 16451860 -
Abstract: PMID: 16688770 -
Abstract: PMID: 16933217 -
Abstract: PMID: 18405364 -
Abstract: PMID: 17919832 -
Abstract: PMID: 19808787 -
Abstract: PMID: 15065184 -
Abstract: PMID: 18182338 -
Abstract: PMID: 18771985 -
Abstract: PMID: 18182339 -
Abstract: PMID: 18563447 Franke B, Vermeulen S, Steegers-Theunissen R, Coenen M, Schijvenaars M, Scheffer H, den Heijer M, Blom H. An association study of 45 folate-related genes in spina bifida: involvement of cubilin (CUBN) and tRNA aspartic acid methyltransferase 1 (TRDMT1). Birth Defects Research (Part A). 2009;85(3):216-26. -
Abstract: PMID: 19319979 -
Abstract: PMID: 17409324 -
Abstract: PMID: 17479212 -
Abstract: PMID: 17486595 -
Abstract: PMID: 17336564 -
Abstract: PMID: 17024475 Centers for Disease Control and Prevention (CDC). Racial/ethnic differences in the birth prevalence of spina bifida: the United States, 1995-2005. MMWR Morb Mortal Wkly Rep. 2009 Jan 9;57(53):1409-13. Abstract: PMID: 19129744
-
Abstract: PMID: 18655127 -
Abstract: PMID: 11717624 -
Abstract: PMID: 12673636 Verbeek EJ, vd Hoeven JH, Maurits NM, Sollie KM, Bos AF, Brouwer OF, den Dunnen WF, Sival DA. Muscle echogenicity is increased in fetuses with spina bifida aperta. Cerebrospinal Fluid Research. 2007 Dec;4(Suppl 1):S11. -
Abstract: PMID: 15286226 -
Abstract: PMID: 19447572 -
Abstract: PMID: 19519395 -
Abstract: PMID: 14710103 -
Abstract: PMID: 12614945 -
Abstract: PMID: 18212354 -
Abstract: PMID: 15172003 -
Abstract: PMID: 18538144 -
Abstract: PMID: 15613950 -
Abstract: PMID: 16280367 -
Abstract: PMID: 16933125 -
Abstract: PMID: 19228313 -
Abstract: PMID: 16199631 -
Abstract: PMID: 16769022 -
Abstract: PMID: 17154227 Acharya G, Jonsrud C, Maltau JM. Neural tube defect: issues of prenatal diagnosis, genetic counselling, prevention, and management. Ultrasound. 2006 May;14(2):95-99. -
Abstract: PMID: 16387462 -
Abstract: PMID: 16199631 Acharya G, Jonsrud C, Maltau JM. The neural tube defect: issues of prenatal diagnosis, genetic counselling, prevention, and management. Ultrasound. 2006 May;14(2):95-99. -
Abstract: PMID: 19301349 -
Abstract: PMID: 10517457 -
Abstract: PMID: 17086581 -
Abstract: PMID: 18598123 -
Abstract: PMID: 17437193 -
Abstract: PMID: 15008380 -
Abstract: PMID: 16978979 -
Abstract: PMID: 18096728 -
Abstract: PMID: 19106353 -
Abstract: PMID: 19643781 -
Abstract: PMID: 19244067 -
Abstract: PMID: 17260216 -
Abstract: PMID: 17710873 -
Abstract: PMID: 17554800 Kucinqska-Chahwan A, Roszkowski T, Debski R. Spina bifida: retrospective analysis of fetuses diagnosed in the Department of Obstetrics and Gynecology of the Postgraduate Center of Medical Education between 1997 & 2004. Ginekol Pol. 2006 Jan;77(1):10-6. Abstract: PMID: 16736955
Zhang XL, Wang LX, Sun CK. Diagnosis of spina bifida in fetuses using tomographic ultrasound imaging: a preliminary study. Nan Fang Yi Ke Da Xue Xue Bao. 2008 May;28(5):832-4. Abstract: PMID: 18504213
-
Abstract: PMID: 11202581 -
Abstract: PMID: 15517549 -
Abstract: PMID: 16846520 -
Abstract: PMID: 16470735 Mehta TS, Levine D. Ultrasound and MR imaging of fetal neural tube defects. Ultrasound Clinics. 2007 April;2(2):187-201. -
Abstract: PMID: 16971596 -
Abstract: PMID: 17032846 -
Abstract: PMID: 15700225 -
Abstract: PMID: 16619377 -
Abstract: PMID: 15914682 -
Abstract: PMID: 18762395 -
Abstract: PMID: 17710413 Resta M, Burdi N, Donatelli M, Resta M. Fetal magnetic resonance imaging in holoprosencephaly, agenesis of the corpus callosum, and Chiari II malformation. Rivista di Neuroradiologia. 2006;19:13-18. -
Abstract: PMID: 17874087
1
3
4
8
9
24
31
35
51
54
69
70
75
83